Study Design Resources

Study Design Resources
Study Design Resources
Steps
- Choosing a Research Question
- Formulating Hypotheses
- Identifying/Selecting Endpoints or Outcomes
- Choosing a Study Design
Research Question
A well thought out and specific research question paired with an appropriate study design is crucial to obtaining meaningful results.
Formulating a specific and concise research question can be complicated and will often be a multi-step process. We encourage collaboration with a statistician early and often to ensure the research question is specific and answerable through data collection and statistical analyses.
The below table gives a brief example of this process
Research Questions
Key Step
Example
Patient Problem
How should I describe a group of people?
Children under the age of 12 have condition X
Exposure
What is the main course of treatment I should consider?
If I add drug X into their therapy regimen, what will occur?
Comparison
What other treatment options are available?
What occurs if I increase their current dosage of drug X
Outcome
What are the patient’s expectations?
Patients want increased symptom control and minimal side effects
SOURCE
Formulating Hypotheses
Along with a specific and concise research question, hypothesis questions will need to be formed. Research questions in nature are more inquisitive or interrogative. Hypotheses are more predictive and are usually made with preexisting knowledge of the subject. Hypotheses should always be written declaratively instead of interrogatively, like a research question. A hypothesis statement should be developed prior to any data collection.
Do you need help distinguishing between research questions and hypotheses? Visit the following link for in text descriptions of each and a video distinguishing the characteristics between the two provided by Center for Innovation in Research and Testing.
The following link from UCLA provides a nice introduction to hypothesis testing.
Identifying/Selecting Endpoints or Outcomes
Endpoints need to be established immediately after forming a research question as is steers the user in the right direction when establishing whether an outcome will be quantitatively or qualitatively determined.
Study Design
Choosing the Correct Study Design
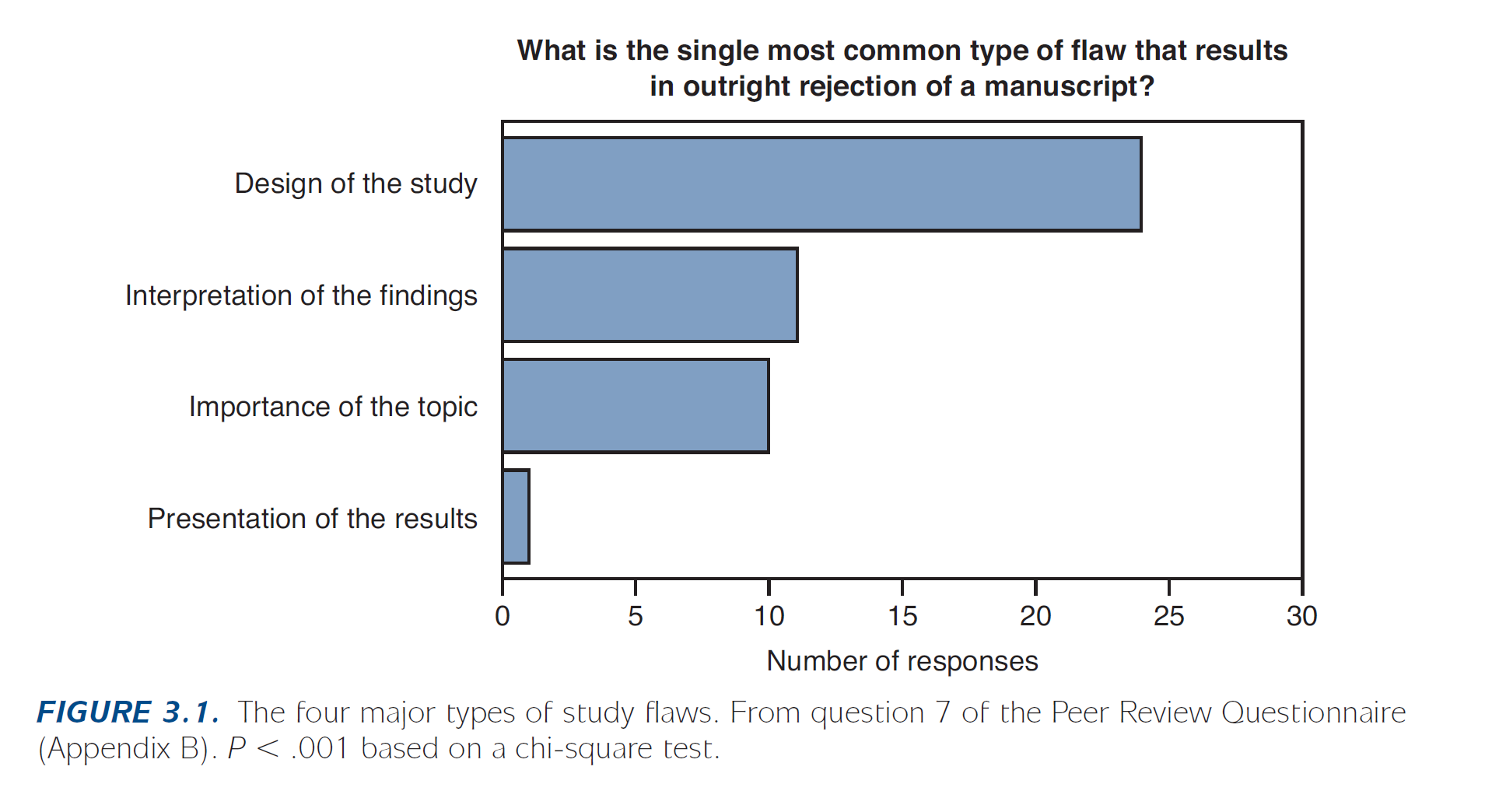
It is critical to choose the correct study design for your question to obtain meaningful results. Because some research questions can often be answered by more than one method/design, the choice of design depends on a variety of considerations, including: availability of time, resources, and data. We encourage a discussion with a statistician early in the research process to ensure a successful study design plan. According to Byrne (Byrne 2017 – Publishing your medical research), study design is the single most common type of flaw that results in outright rejection of a submitted manuscript.
And more importantly, a flawed design may lead to spurious results that cannot answer your research questions.
More Information on Outcomes:
Study Design Reading List
Designing Clinical Research by Hulley et al. (2013)
Fundamentals of Clinical Trials by Friedman et al. (2015)
Evaluating Clinical and Public Health Interventions: A Practical Guide to Study Design and Statistics by Katz (2010)
Basic Statistics for the Health Sciences by Kuzma and Bohnenblust (2004)
How to Write a Lot: A Practical Guide to Productive Academic Writing by Silvia (2007)
Medical Uses of Statistics by Bailar and Hoaglin (2009)
Statistics with Confidence: Confidence Intervals and Statistical Guidelines by Altman et al. (2000)
How to Report Statistics in Medicine: Annotated Guidelines for Authors, Editors, and Reviewers by Lang and Secic (2006)
The Man Who Discovered Quality: How W. Edwards Deming Brought the Quality Revolution to America – The Stories of FORD, XEROX, and GM by Gabor (1992)
Epidemiology by Gordis (2013)
Essentials of Medical Statistics by Kirkwood and Sterne (2003)
Clinical and Translational Science: Principles of Human Research by Robertson and Williams (2016)
Statistical Modeling for Biomedical Researchers: A Simple Introduction to the Analysis of Complex Data by Dupont (2009)
Modern Epidemiology by Rothman et al. (2012)
Clinical Prediction Models: A Practical Approach to Development, Validation, and Updating by Steyerberg (2010)
Encyclopedia of Biostatistics: 8-Volume Set by Armitage and Colton (2005)
Essentials of Writing Biomedical Research Papers by Zeiger (1999)
Statistical Issues in Drug Development by Senn (2008)
Experimental Design for Biologists by Glass (2014)
Experimental Design for the Life Sciences by Ruxton and Colegrave (2010)
Thinking, Fast and Slow by Kahneman (2013)
Resources:
A brief guide to the different study types and a comparison of advantages and disadvantages
An overview of study design options
Types of clinical study designs:
A review of novel methods and technologies for 21st-century clinical trials
Research designs in sports physical therapy
Alternative and Emerging Study Designs
Contact Us
Williams Building
University of Utah Research Park
Williams Building, 1st floor
295 South Chipeta Way
Salt Lake City, Utah
Map
Parking: During construction, you may park on the bottom floor of the south parking structure.
Contact
Camie Derricott
Phone: 801-587-5212
Fax: 801-581-3623
Acknowledging the SDBC
Please use the following text to acknowledge the CTSI Study Design and Biostatistics Center:
"This investigation was supported by Translational Research: Implementation, Analysis and Design (TRIAD), with funding in part from the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UM1TR004409. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health."
"This investigation was supported by the Study Design and Biostatistics Center (SDBC), with funding in part from the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UM1TR004409. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health."