Grant Writing

Grant Writing
One of the primary missions of the SDBC/PHR is to provide support in the grant application process. Meeting with your SDBC/PHR collaborator early and often will increase the chances for a successful grant application. We recommend that your first meeting occur at least 2-3 months prior to your grant submission deadline.
The following are key areas in which a SDBC/PHR Collaborator can provide guidance throughout the grant writing process.
- Clarifying the research questions or hypothesis. Are the primary hypotheses clearly stated, realistic, and testable?
- Identifying the outcome variables related to the research questions. Are the primary and secondary end points (if applicable) clearly defined?
- Does the study design appropriately and adequately address the proposed hypothesis?
- Are the issues of bias, blinding, or stratification properly handled in the study?
- Are the assumptions used for sample size estimation clearly elaborated and supported by proper preliminary data and/or references?
- Is there a clearly specified, appropriate data analysis plan?
- For the prospective studies, is there an appropriate data and safety monitoring plan, interim analysis plan, or preestablished early stopping rule?1
The topics and resources in this section are intended as a guide. The funding climate is constantly changing and agencies may change grant application requirements. A perfect grant application may be rejected because incorrect forms or formatting were used.
Your first stop should always be the guidelines provided by the funding agency.
Overview of Grant Writing Process and Getting Started
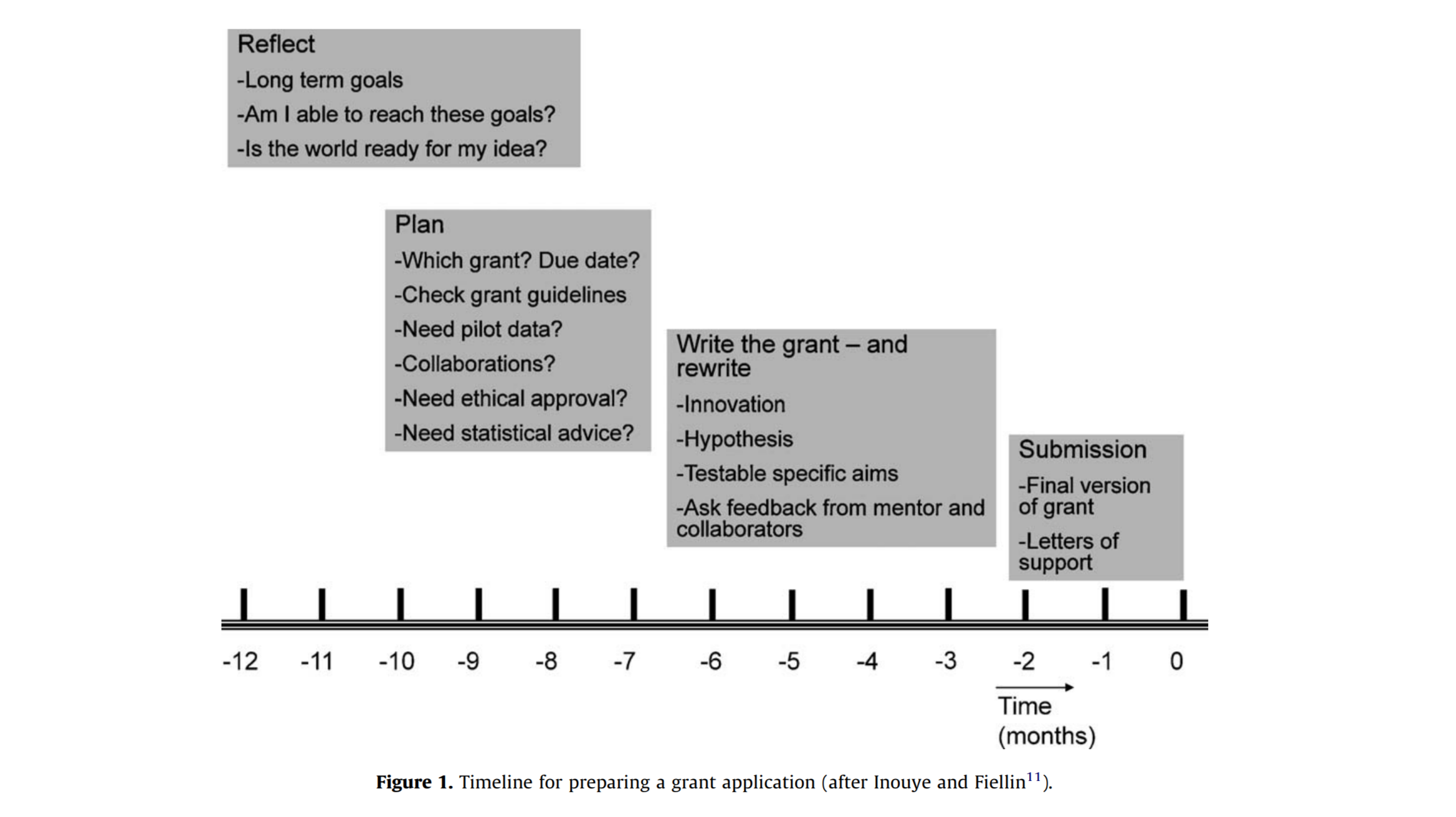
How much time do I need?
Short answer is twice as much time as you think you need. The following guideline to the elements of a grant submission should help you plan your time wisely.
Figure from: Koppelman GH, Holloway JW, Successful grant writing. Paediatric Respiratory Reviews 13 (2012) 63–66. - https://www.sciencedirect.com/science/article/pii/S1526054211000157?via%3Dihub
What are the steps in writing a grant and why does it take so much time?
Table from: Experimental study design and grant writing in eight steps and 28 questions. Georges Bordage, Beth Dawson. - https://onlinelibrary.wiley.com/doi/epdf/10.1046/j.1365-2923.2003.01468.x
REFERENCES:
- Adams-Huet B, Ahn C Bridging Clinical Investigators and Statisticians: Writing the Statistical Methodology for a Research Proposal. Journal of Investigative Medicine Volume 57, Number 8, December 2009 Writing the Statistical Analysis Plan https://sjoseph.ucdavis.edu/proposalwritingandfundingresearch/manuals-and-guides-1
Our Office
Williams Building
University of Utah Research Park
Williams Building, 1st floor
295 South Chipeta Way
Salt Lake City, Utah
Map
Parking: During construction, you may park on the bottom floor of the south parking structure.
Contact
Camie Derricott
Camie.Derricott@hsc.utah.edu
Acknowledging the SDBC
Please use the following text to acknowledge the CTSI Study Design and Biostatistics Center:
"This investigation was supported by Translational Research: Implementation, Analysis and Design (TRIAD), with funding in part from the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UM1TR004409. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health."
"This investigation was supported by the Study Design and Biostatistics Center (SDBC), with funding in part from the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UM1TR004409. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health."